

| name | Amanita lavendula |
| name status | nomen acceptum |
| author | (Coker) Tulloss, K. W. Hughes, Rodrig. Cayc., & Kudzma |
| english name | "Coker's Lavender Staining Amanita" |
| images |
  1. Amanita citrina f. lavendula, Coker's photo of lectotype (Coker 1432), Chapel Hill, Orange Co., North Carolina, USA.   2. Amanita citrina f. lavendula, Coker's photo of original material (Coker 590), Chapel Hill, Orange Co., North Carolina, USA. ![Amanita sp-lavendula02, Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2].](image/uploaded/c/citrlavx.jpg) ![Amanita sp-lavendula02, Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2].](image/uploaded/c/citrlavx.jpg) 3. Amanita sp-lavendula02, Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2]. ![Amanita sp-lavendula02,' Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2] with hypermagenta test.](image/uploaded/c/citrlavm.jpg) ![Amanita sp-lavendula02,' Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2] with hypermagenta test.](image/uploaded/c/citrlavm.jpg) 4. Amanita sp-lavendula02,' Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2] with hypermagenta test. ![Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8].](image/uploaded/s/sp-lavendula-2_178456wb.jpg) ![Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8].](image/uploaded/s/sp-lavendula-2_178456wb.jpg) 5. Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8]. ![Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8].](image/uploaded/s/sp-lavendula-2_178458wb.jpg) ![Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8].](image/uploaded/s/sp-lavendula-2_178458wb.jpg) 6. Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8]. |
| intro | For the time being, please, see the technical tab for this page. |
| discussion |
The present species was originally described from
North Carolina, USA, where it occurs commonly in
pine (Pinus) forest and mixed woods
including pine and oak (Quercus).
Within its range, it and its two look-alikes
(A.
cornelihybrida and
A.
americitrina) are
among the last species of
Amanita to be found at the end of the
mushroom season—in autumn or early
winter. This species has been mistakenly referred to "A. citrina" (≡A. mappa). The original description of the present taxon includes some material on which no lavender staining was seen according to Coker's annotation on the herbarium packets. RET's personal experience indicates that, without cold nights, many specimens of this mushroom show no lavender at all. Hence, it is possible that Coker's collections include some material that might better be included under one of the two code named taxa (above). Those taxa have larger and rounder spores than those found in A. lavendula. Based on limited experience, the lavender staining seems to RET to be much more common (certainly more widespread on the fruiting body) in specimens collected after a light frost or after a night when the temperature (allowing for windchill) has been below freezing. This suggests that environmental factors (presumably low temperature) are correlated with the accentuation of lavender tinted regions and lavender staining on freshly cut surfaces. 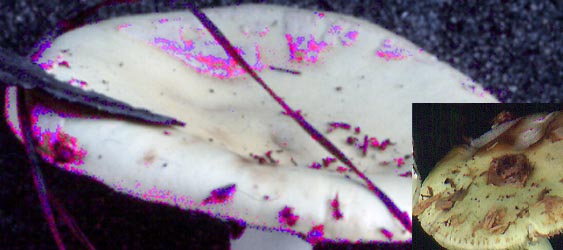
As an experiment (using Adobe Photoshop®), we increased the saturation of magenta to the maximum in a picture of a mushroom of "the A. lavendula group" from near the South Carolina-Georgia border and did the same for a picture of a yellow capped specimen of A. mappa (Batsch) Fr. from England. The combined illustrations are shown immediately above. Even at high magnification, the European specimen displayed very little magenta spotting and the North American specimen, as can be seen, has extensive areas of magenta and colors in which magenta is a significant component, especially in association with volva patches. Both pictures were taken on Kodachrome ASA 64 slide film with a Canon AE-1 camera in autumn or early winter (although at different latitudes). The pronounced difference is noticeable despite the fact that the film is not terribly sensitive to the lavender tints and failed to reproduce them well in photographs taken of specimens in which purplish lavender was a component of cap skin coloring. In addition, the lack of lavender was also present on A. mappa caps that were less mature than the one pictured and in which the volva had not darkened so distinctly. There was no magenta visible in the flesh of sectioned fruiting bodies of the same taxon. There was also a very minimal reaction to maximum saturation of magenta in the case of white specimens of A. mappa.  We continued the experiment with additional
photographs of other photographs of "A.
lavendula group" fruiting bodies
taken in December at the same South Carolina
site. The same phenomena was
observed—significant areas of the fruiting body
(especially those that had seemed lavender to the
naked eye) became strongly magenta. One of
the photos from the site shows extensive lavender
coloration over most of the cap (first row, right,
at the top of this page). In this case,
nearly the entire pileus became magenta as a result
of the maximum saturation experiment ("hypermagenta
test") (left). To date, the hypermagenta test
has been applied with negative results only to
photographs of citrina-like material
from Europe (France, Norway
and the United Kingdom) and to specimens of the
current taxon on which no lavender could be
detected by the eye. We are very interested
in testing photographs (for which there are dried
voucher specimens) from parts of Europe other than
those cited above.
We continued the experiment with additional
photographs of other photographs of "A.
lavendula group" fruiting bodies
taken in December at the same South Carolina
site. The same phenomena was
observed—significant areas of the fruiting body
(especially those that had seemed lavender to the
naked eye) became strongly magenta. One of
the photos from the site shows extensive lavender
coloration over most of the cap (first row, right,
at the top of this page). In this case,
nearly the entire pileus became magenta as a result
of the maximum saturation experiment ("hypermagenta
test") (left). To date, the hypermagenta test
has been applied with negative results only to
photographs of citrina-like material
from Europe (France, Norway
and the United Kingdom) and to specimens of the
current taxon on which no lavender could be
detected by the eye. We are very interested
in testing photographs (for which there are dried
voucher specimens) from parts of Europe other than
those cited above.
 Recently collected material (2006, from North Carolina and Tennessee and showing the strongest lavender color RET has ever seen in the taxon) photographed with a digital camera (Olympus C-5500 Zoom) produced photographs that reacted to the hypermagenta test when there were distinct regions of lavender on the cap, the stem, or on exposed flesh (at right). Research on fruiting body chemistry (proteins) several years ago at the University of Tennessee, Knoxville, was unable to differentiate between lavender staining and non-lavender staining material from Tennessee and North Carolina on the basis of a set of proteins studied. On the other hand, comparison of the forms of hyphae and rate of hyphal growth in culture between a similar pair of sets of specimens showed a strong distinction, in research at the same university. Perhaps these cultures were based on what we now know are distinct species in the "group." more?—R. E. Tulloss and L. Possiel |
| brief editors | RET |
| name | Amanita lavendula | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| author | Tulloss, K. W. Hughes & Rodríg.-Cayc. in Tulloss et al. 2015. Amanitaceae 1(2): 2. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| name status | nomen acceptum | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| english name | "Coker's Lavender Staining Amanita" | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| synonyms |
≡Amanita citrina f. lavendula(Coker) Veselý. 1933. Ann. Mycol. 31(4): 239.
≡Amanita mappa var. lavendula Coker. 1917. J. Elisha Mitchell Scient. Soc. 33(1/2): 39, pl. 22-23, 64.
≡Amanita citrina var. lavendula (Coker) Sartory & L. Maire. 1922. Compend. Hymenomyc.—Amanita ??: 25.
≡Amanita porphyria var. lavendula (Coker) L. Krieg. 1927. Mycologia 19: 309.
≡Amanita brunnescens f. lavendula (Coker) E.-J. Gilbert nom. inval 1941. Iconogr. Myco. (Milan) 27 suppl. 1 (1-3): 336. [Not accepted by author in original publication. ICBN §34.1(a)] The editors of this site owe a great debt to Dr. Cornelis Bas whose famous cigar box files of Amanita nomenclatural information gathered over three or more decades were made available to RET for computerization and make up the lion's share of the nomenclatural information presented on this site. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MycoBank nos. | 508879, 166594, 508880, 171834, 807618 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GenBank nos. |
Due to delays in data processing at GenBank, some accession numbers may lead to unreleased (pending) pages.
These pages will eventually be made live, so try again later.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| lectotypes | NCU | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| lectotypifications |
Tulloss et al. 2015. Amanitaceae 1(2): 2. Rejected lectotypification of Neville and Poumarat (2004: 818) rejected by Tulloss (2005a. Mycotaxon 92: 480 [footnote]). | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| intro |
The following text may make multiple use of each data field. The field may contain magenta text presenting data from a type study and/or revision of other original material cited in the protolog of the present taxon. Macroscopic descriptions in magenta are a combination of data from the protolog and additional observations made on the exiccata during revision of the cited original material. The same field may also contain black text, which is data from a revision of the present taxon (including non-type material and/or material not cited in the protolog). Paragraphs of black text will be labeled if further subdivision of this text is appropriate. Olive text indicates a specimen that has not been thoroughly examined (for example, for microscopic details) and marks other places in the text where data is missing or uncertain. The following morphologcial material not cited as the work of another researcher is based on original research of R. E. Tulloss. Phylogenetic analyses are based on continued original research in the laboratories of Drs. K. W. Hughes (Univ. of Tenn., Knoxville) Dr. L. V. Kudzma (Annandale, New Jersey). All parts of the fruiting body are liable to become lavender or a similar purplish shade when ambient temperature is below ca. 1° C for several hours. This character is shared with the two other known macromorphologically similar taxa in eastern North America—A. cornelihybrida and A. americitrina. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| pileus |
protolog: "...from 3.5 to 8
cm in diameter, flat or slightly depressed in
center (sometimes slightly gibbous in center) a
light but distinct primrose yellow (not the dull
egg yellow shades of A.
russuoloides), often with stains of light
brown, lavender, purple lavender, or a combination
of these; somewhat viscid when damp, shining when
dry"; context "...nearly white, sometimes
quickly turning to shades of lavender when cut,
quite thin, only 2 mm thick at center of gills...";
margin "disctinctly striate when mature, or
the striae may be scarcely visible until the margin
begins to dry," not appendiculate; universal
veil as occasional flat, irregular, lavender or
pink-lavender patches." RET: 32 - 115 mm wide, 4A3 at margin and 3B3 over disc or very pale citrine-yellow except brownish over disc or mostly ivory and tinged with tan over disc, becoming paler (except over disc) with age, planoconvex with slight umbo to planar, sometimes deeply rimose, tacky to wasy, shiny; context 3 - 5 mm thick, white to grayish white, sometimes with watersoaked line above lamellae and above join with stipe context, thinning evenly to margin; margin not striate, not appendiculate; universal veil absent or in scattered flat patches, 5B2 to grayish. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| lamellae |
protolog: "...pure white, free
but close to the stem and connected by a line which
runs down a little way [on the apical region of the
stipe], deepest near their middle, where they were
from 3.5 to 7 mm deep [i.e., broad], deep according
to the size of the plant [sic]..., none forked.";
lamellulae "..many...." RET: free, without decurrent line at stipe apex, close to crowded, sordid white to cream to ca. 3A2 in mass, white to pale watersoaked cream and sometimes becoming slightly sordid in age in side view, 2.5 - 6.5 mm wide, broadest ca. midlength; lamellulae subtruncate to truncated (shortest) or subattentuate to attenuate (longest). | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| stipe |
protolog: ?...up to 10 cm
long and from 6 to 10 mm thick in center, smooth
and somewhat silky-shining, faint primrose-yellow
[appearing to be of substance continuous with
partial veil, which breaks up and remains
concolorous with partial veil, but fades] above and
nearly white below the veil, but often with cream,
brown, or lavender tints, and brown where bruised";
context "solid but sometimes nearly hollow
from the separation of the looser central fibers,
no distinct central cylinder"; bulb “"large
and abrupt, but variable, sometimes 2.8 cm in
diameter, soft and spongy generally with an
abruptly truncated top, which may be quite smooth
or show slight marginal projections representing
the volva"..."the surface is a distinct lavender
color, sometimes pinkish or brownish lavender or
rarely nearly white (as in No. 1399, but even in
this collection the volva patches on the cap were
lavender), internally it is white"; partial
veil "primrose yellow, thin, delicate, but not
flocculent or friable, the lower side often showing
the fibers by which it was attached to the
stem...it remains attached to the stem from 2 to
3.5 cm below the cap, generally collapsing tightly
against the stem, and so delicate at times that it
is scarcely noticeable on the mature plant [sic]
except where its free edge marks out a colored line
against the stem. At other times the veil remains
expanded for some time as a perfect skirt, and is
quite perfect in the mature plant [sic]";
universal veil "there is no volval
cup." RET: 80 - 163 × 6 - 17 mm, white below partial veil, pale citrine-yellow above, becoming grayish with age or handling, rusty brown in old wounds, narrowing upward (sometimes only slightly) or cylindric, slightly to markedly flaring at apex, pruinose and similar to upper surface of partial veil above latter, below satiny and striatulate at first, sometimes becoming subshaggy-fibrillose at least in lower third of stipe; bulb 10 - 23 × 13 - 24 mm, subglobose, sometimes with rusty stains on exterior; context stuffed or solid, off-white, with rusty marks in old wounds, with central cylinder 4 - 8.5 mm wide and sometimes becoming watersoaked, insect tunnels not observed; partial veil membranous, persistent, skirt-like, superior, pale yellow, becoming grayish from edge inward with age, faintly striate above (at least near edge), bottom surface concolorous with upper surface and bearing subrectangular fragments of graying universal veil limbus internus around the edge; universal veil as brief and irregular circumcissile limb separated from stipe by sinus ca. 2± mm wide, white at least at first, with color change similar to that on pileus. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| odor/taste |
protolog: Odor "...when
freshly cut like raw green peanuts." RET: Odor like freshly dug potatoes. Taste not recorded. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| macrochemical tests |
Spot test for tyrosinase (0.1% paracresol) - negative throughout basidiome for both youngest and oldest specimens tested. Spot test for tyrosinase (L-tyrosine) - negative throughout basidiome for specimen just prior to maturity. Test vouchers: Tulloss 11-30-85-E, Tulloss 7-18-96-F, 9-26-97-D. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| lamella trama | bilateral, divergent; ??. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| subhymenium | pseudoparenchymatous (cellular); ??. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| basidia | ?? × ?? μm, dominantly 4- and infrequently 1- or 2-sterigmate, with sterigmata ?? × ?? μm; clamps not observed. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| stipe context | longitudinally acrophysalidic: ??. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| lamella edge tissue | sterile; ??. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| basidiospores |
composite data from original material, RET:
[40/2/2] (5.8-) 6.0 - 7.5
(-8.1) × (5.1-) 5.2 - 6.5 (-7.0) μm, (L=
6.4 - 6.8 μm; L' = ?? μm; W = 5.8 -
6.0 μm; W' = ?? μm; Q = (1.03-) 1.05 - 1.22
(-1.25); Q = 1.11 - 1.13; Q' = ??),
??, ??, thin-walled, smooth, amyloid, subglobose to
broadly ellipsoid, adaxially flattened;
apiculus sublateral, cylindric, varying in
width; contents granular; white in
deposit.
composite data from all collections revised by RET & CRC: [140/7/6] (5.8-) 6.0 - 8.0 (-9.7) × (4.9-) 5.3 - 7.0 (-8.1) μm, (L = 6.5 - 7.2 μm; L' = 6.9 μm; W = 5.8 - 6.4 μm; W' = 6.1 μm; Q = (1.02-) 1.05 - 1.29 (-1.49); Q = 1.10 - 1.18; Q' = 1.13), hyaline, colorless, thin-walled, smooth, amyloid, subglobose to broadly ellipsoid, occasionally globose, infrequently ellipsoid, predominantly adaxially flattened; apiculus sublaterial, cylindric; contents predominantly guttulate, occasionally granular; white in deposit. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ecology | Subgregarious to solitary. La Vega, Dominican Republic: Under Pinus occidentalis. Hidalgo, México: In friable dark soil of mesophytic Quercus-Pinus forest (at least sometimes Pinus patula). Connecticut: In loam and plant litter of deciduous forest with Quercus alba, Q. rubra, Betula sp., Fagus grandifolia, etc. Missouri: A pair. At 262 m elev. New Jersey: Clustered to gregarious. At 35 m elev. In deep sandy soil of Pinus rigida-Quercus barrens or in similar soil under Pinus. South Carolina: In loamy soil of Pinus-Quercus forest. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| material examined |
CANADA:
ONTARIO—Norfolk Co., Port Dover
[42.7863° N/ 80.198° W, 182 m], 1.ix.2013 Eva
Skific s.n.
[mushroomobserver.org
#144353]
(RET 566-3, nrITS seq'd.); Walsh
[42.7616° N/ 80.394° W, 223 m], 28.ix.2013 E.
Skific s.n. [mushroomobserver.org
#146683]
(RET 575-6, nrITS seq'd.), 3.viii.2014 Eva
Skific s.n. [mushroomobserver
#172318]
(RET 639-7, nrITS & nrLSU seq'd.).
DOMINICAN REPUBLIC:
LA VEGA—N of Buena Vista, 4-5 km N
of Jarabacoa, along Nat. Rte. 28, 10.xi.2011 C.
Angelini ANGE95 (RET 693-2,
nrITS-LSU seq'd.).
MÉXICO: HIDALGO—Mpio.
Molango - Laguna Atezca [20°47’32” N/ 98°44’53” W,
1260 m], 18.vii.1996 Dr. Arturo Estrada Torres &
Lucía Varela Fregoso s.n. [Tulloss 7-18-96-H] (RET
253-2, nrITS seq'd.). Mpio. unkn. - ca.
northern jct. of Méx. Rte. 105 deviat. for
Zacualtipan & main Rte. 105 [20°40’28” N/
98°40’29” W], 18.vii.1996 Adriana Montoya Equivel,
A. Estrada Torres, L. Varela Fregoso & R. E. Tulloss
[Tulloss 7-18-96-E] (RET 252-7, nrITS seq'd.),
[Tulloss 7-18-96-F] (RET 252-10, nrITS seq'd.).
U.S.A.:
CONNECTICUT—Middlesex Co. - East Haddam,
Devil's Hopyard St. Pk. [41°28’57” N/ 72°20’30” W],
21.ix.1996 R. E. Tulloss 9-21-96-G (RET 250-3,
nrITS seq'd.).
MISSOURI—Ste. Genevieve Co. - W of Ste.
Genevieve, Hawn St. Pk. [37.8337° N/ 90.2416° W,
262 m], 30.x.2011 Patrick Harvey s.n.
[mushroomobserver.org #81042]
(RET 495-8, nrITS seq'd.), 14.x.2012 P. Harvey s.n.
[mushroomoberver.org #113456]
(RET 554-9, nrITS & nrLSU seq'd.),
[mushroomoberver.org #113460]
(RET 554-7, nrLSU seq'd.),
[mushroomoberver.org #113461]
(RET 554-8, nrLSU seq'd).
NEW JERSEY—Burlington Co. - Brendan T.
Byrne | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| discussion |
A comparison of sporographs based on the original
material of the present taxon that are candidates
for designation as its lectotype and
Amanita brunnescens var.
straminea, a clearly citrinoid taxon,
are found in the following diagram:Note regarding lectotypification: Thirteen of 17 syntypes for A. mappa var. lavendula were located by RET in NCU. These were examined in order to support selection of a lectotype from among them. In the process of selecting a lectotype, RET first eliminated material for which the annotation of fresh material does not unambiguously state that lavender was present on a basidiome in a given collection. In addition, a syntype was eliminated from consideration if it is missing some significant element such as universal veil on the bulb or the partial veil. Existence of a photograph of the fresh material and, especially, publication of such a photograph of a given collection in the protolog was viewed as increasing the value of the collection, all other elements being equal. When it became clear that the two spore size ranges cited by Coker were associated with different spore shapes and that the size range provided by Coker in his basic description (rather than in a comment) was associated with the best candidate for lectotype, this was the final deciding factor in choosing a collection to propose as lectotype (Tulloss et al 2015). Note regarding molecular results (1.vi.2015 per Hughes and Tulloss, personal communication): Recent molecular studies indicate that collections previously determined as A citrina in the sense of eastern U.S. authors or A. citrina var. lavendula can be divided into three distinct taxa (segregated by nrITS and nrLSU sequences). Note: The illustration of the collection from North Carolina (Tulloss 9-30-06-C) indicates that a fruiting body with rather strong yellow on the cap (as opposed to the pallid cap described by Coker) can have strong lavender staining and be assignable to A. lavendula. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| citations |
Those working on this species acknowledge the support
of the University of Tennessee, Knoxville, and
National Science Foundation grant DEB 1144974.—R. E.
Tulloss and K. W. Hughes.Remaining unsorted collections. Attention required: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| editors | RET | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Information to support the viewer in reading the content of "technical" tabs can be found here.
| name | Amanita lavendula |
| name status | nomen acceptum |
| author | (Coker) Tulloss, K. W. Hughes, Rodrig. Cayc., & Kudzma |
| english name | "Coker's Lavender Staining Amanita" |
| images |
  1. Amanita citrina f. lavendula, Coker's photo of lectotype (Coker 1432), Chapel Hill, Orange Co., North Carolina, USA.   2. Amanita citrina f. lavendula, Coker's photo of original material (Coker 590), Chapel Hill, Orange Co., North Carolina, USA. ![Amanita sp-lavendula02, Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2].](image/uploaded/c/citrlavx.jpg) ![Amanita sp-lavendula02, Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2].](image/uploaded/c/citrlavx.jpg) 3. Amanita sp-lavendula02, Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2]. ![Amanita sp-lavendula02,' Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2] with hypermagenta test.](image/uploaded/c/citrlavm.jpg) ![Amanita sp-lavendula02,' Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2] with hypermagenta test.](image/uploaded/c/citrlavm.jpg) 4. Amanita sp-lavendula02,' Wildacres Retreat, ca. Little Switzerland, McDowell Co., North Carolina, USA [RET 396-2] with hypermagenta test. ![Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8].](image/uploaded/s/sp-lavendula-2_178456wb.jpg) ![Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8].](image/uploaded/s/sp-lavendula-2_178456wb.jpg) 5. Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8]. ![Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8].](image/uploaded/s/sp-lavendula-2_178458wb.jpg) ![Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8].](image/uploaded/s/sp-lavendula-2_178458wb.jpg) 6. Amanita sp-lavendula02, Hawn St. Pk., Ste. Genevieve Co., Missouri, USA [RET 495-8]. |
| photo |
Dr. William Chambers Coker - (1) Chapel Hill,
Orange County, North Carolina, U.S.A.
[Image included in protolog.] RET (2-3) Wildacres Retreat, ca. Little Switzerland, McDowell County, North Carolina, U.S.A. [RET 396-2]. Patrick Harvey (4-5) Hawn State Park, Ste. Genevieve County, Missouri, U.S.A. Original size images and additional images of the same collection are on www.mushroomobserver.com #81042. |
Each spore data set is intended to comprise a set of measurements from a single specimen made by a single observer; and explanations prepared for this site talk about specimen-observer pairs associated with each data set. Combining more data into a single data set is non-optimal because it obscures observer differences (which may be valuable for instructional purposes, for example) and may obscure instances in which a single collection inadvertently contains a mixture of taxa.


